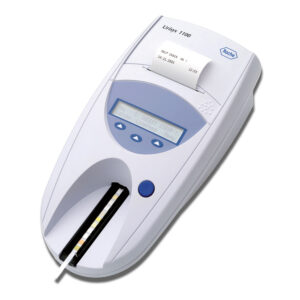
CLINITEK Status® Connect System
CLINITEK Status® Connect System simplifies wireless or wired connectivity and testing oversight in point-of care urinalysis for improved risk management.
- Offers flexible connectivity solutions by integrating data directly to the LIS, EMR, or via point-of-care data management software solutions
- Provides improved POC testing workflow efficiencies when interfaced to leading data management solutions
- Improved barcode response time allows quick entry of lot and expiration data with a single scan
- Improves risk management through advanced operator control functions, prevents unauthorized use
- Drives compliance across testing sites with programmable QC protocols and QC lockout
- Auto-Checks* help to ensure the quality and accuracy of data while facilitating an enhanced interpretation of results
£439.00
2 in stock
Additional information
CLINITEK Status® Connect System
CLINITEK Status® Connect System simplifies wireless or wired connectivity and testing oversight in point-of care urinalysis for improved risk management.
- Offers flexible connectivity solutions by integrating data directly to the LIS, EMR, or via point-of-care data management software solutions
- Provides improved POC testing workflow efficiencies when interfaced to leading data management solutions
- Improved barcode response time allows quick entry of lot and expiration data with a single scan
- Improves risk management through advanced operator control functions, prevents unauthorized use
- Drives compliance across testing sites with programmable QC protocols and QC lockout
- Auto-Checks* help to ensure the quality and accuracy of data while facilitating an enhanced interpretation of results
Features & Benefits
- Centralize control over your point-of-care urinalysis testing program with the CLINITEK Status Connect
Supports Flexible & Efficient Connectivity
- Connect to your hospital’s LIS/HIS, EMR or point-of-care middleware solution
- Connects by wireless, Ethernet or serial transmission
- Supports HL7 and POCT1-A2 protocols
-
Interfaces with leading point-of-care data management solutions for centralized control and testing oversight, including:
- Siemens’ RAPIDComm® Data Management System and Connectivity Solution
- Conworx Technology POCcelerator™
- MAS RALS®-Plus
- TELCOR Quick-Linc®
Advanced Operational Control and Management
- Provides customized operator access level
- Supports operator ID entry and lockout
- Prevents unauthorized use, based on customizable settings
Enhanced Quality Control
- Optional barcode reader eliminates manual steps and reduces transcription errors
- Maintain up-to-date operator certification records and restrict operator access as needed
- Define specific QC protocols for operators across multiple sites
- QC lockout if protocol is not followed or QC test fails
- QC documentation and report downloads are available on demand
- Store up to 200 QC test records
Improved Data Integrity
-
Auto-Checks:
- Checks each strip for humidity to prevent reporting of falsely elevated results
- Detects interferences in test samples and provides printed notation for improved results interpretation†
- Identifies Siemens strip type to save time
Assays
The CLINITEK Status Connect System provides automated reading of the Multistix® family of urinalysis tests:
- Leukocyte
- Nitrite
- Protein
- Blood
- Glucose
- Ketone
- Bilirubin
- Urobilinogen
- pH
- Specific Gravity
- Creatinine‡
- Protein-to-Creatinine Ratio‡
- Albumin
- Albumin-to-Creatinine Ratio (ACR)
- CLINITEST® hCG Pregnancy Test
Semi-quantitative Tests Measured |
|
|---|---|
| Routine urine tests | albumin, bilirubin, creatinine, glucose, ketone, leukocytes, nitrite, pH, protein, specific gravity, and urobilinogen |
| Kidney disease test | albumin-to-creatinine ratio |
| Pregnancy test | Human Chorionic Gonadotropin (hCG) |
General Specifications |
|
| Depth | 10.7 in. or 27.2 cm |
| Width | 6.7 in. or 17.1 cm |
| Height | 7.5 in. or 18.5 cm |
| Weight | 5.0 lbs or 2.3 kg |
| Electrical rating | 100V - 240V AC, 50-60 Hz (with in-line lead) |
| Battery operation | Not available |
| Ambient temperature range | 18 to 30ºC (64 to 86ºF) |
| Humidity range | 18-80% relative humidity, non-condensing |
| Compliance | UL, CE, EMC |
| Calibration | automatic, self-calibrating |
Communication Capabilities |
|
| Communication protocol | HL7, POCT1-A2 |
| Data transmission options | wireless, Ethernet or serial connection |
| Interfaces with leading point-of-care data management solutions |
Siemens’ RAPIDComm® Data Management and Connectivity Solution MAS RALS®-Plus Telcor Quick-Linc® POCcelerator™ |
Memory |
|
| Patient test results | 700 |
| QC test results | 200 |
| Operator IDs | 700 |
Quality Control Management Features |
|
| QC testing and reporting | QC reminders or mandatory prompt, QC lockout |
Barcode Data Entry |
|
| Supported data entry | Patient ID, operator ID and name, QC control type, lot number, and expiration date |
Barcode Symbology Supported |
|
| Supported symbology | Code 93, Code 39 (with and without check digit), Code 128, Coda Bar, Interleaved 2 of 5 (with and without check digit) |
Operator Security Features |
|
| Supported operators | Define up to 700 operators |
| Security features |
Operator lock-out to prevent unauthorized use Customize access level for each operator for the following functions:
|
We also recommend
Supplying Medical Equipment to the NHS for over 20 years
With excellent service and after sale support. Please dont hesitate to contact us.